 
The Lewis structure indicates that each Cl atom has three pairs of electrons that are not used in bonding. Show Electronic configuration of Chlorine. Two chlorine symbols, each surrounded by seven. On which block Chlorine element is present? ĩ) It is Generally use as an solvent for chemical reaction 10) Chlorine is main constituents of extraction of bromin chloroform and carbon tetra chlorideħ) It is use as an disinfectant to kill bacteriaĨ) It is mostly use in industries for make dyes,textiles ,paints. In chlorine 2 electron place in K shell, 8 electrons place in L shell and 7electrons place in M shellģ) Valency : Chlorine show 1 valency In outer shell required only 1 electron to complete octateĤ) Atomic radius : Atomic radius of chlorine is 79 pmĥ) Reactivity : it is more reactive than fluorineģ) It is generally use to make poly vinyl chloride polymer 4) It is mostly use in chemical weaponsĥ) It is use as a oxidising agent in substitution reactionĦ) It is use to made anaesthesia i.e. In this isotopes 17 proton, 17 electron and 19 neutron is present It is present 24% in natureĮlectronic configuration of chlorine is 1S 2 2S 2 2P 6 3S 2 3P 5 Electronic configuration in concert of shell is 2,8,7 It is toxic in natureģ5 Cl : atomic weight of this isotope is 34.9 In this isotope 17 proton, 17 electron and 18 neutron It is present 75% in natureģ7Cl : atomic weight of this isotope is 36.96. It is place in periodic table at 17 th column ( group) and 3 rd row ( period) It is place in halogen familyĦ) Colour : chlorine gas show yellow green colourħ) Odour It odour like choking. 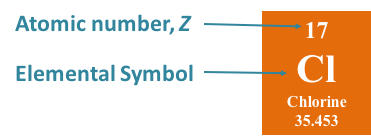 
At ordinary temperature and pressure, chlorine is a pale. It is a member of the halogen group of elements, appearing between fluorine and bromine moving down the periodic table. The name chlorine is originated from Greek word ‘chlorous’ Meaning of this word is ‘ greenish yellow’Ītomic number of chlorine is 17 In chlorine 17 proton and 17 electron is presentĪtomic weight of chlorine is 35.25 In chlorine atom 17 proton and 18 neutron is present hence chlorine show atomic weight is 35.25 Chlorine is a chemical element with atomic number 17 and element symbol Cl. Chlorine is a chemical element of the periodic table with chemical symbol Cl and atomic number 17 with an atomic weight of 35.446 u and is classed as nonmetal and is part of group 17 (fluorine group). It Is discovered by Carl Wilhelm scheele in 1774 And it is in between fluorine and bromine. Its properties are lies between fluorine and bromine. Atomic Mass, Number, Physical, Chemical properties, Electronic configuration, Valency, Chemical reaction, UsesĬhlorine is an non metallic gas. Chlorine – Learn all details regarding Chlorine in Periodic Table i.e. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
